A Role for Pd(IV) in Catalytic Enantioselective C–H Functionalization with Monoprotected Amino Acid Ligands under Mild Conditions
R. Erik Plata, David E. Hill, Brandon E. Haines, Djamaladdin G. Musaev, Ling Chu, David P. Hickey, Matthew S. Sigman, Jin-Quan Yu, and Donna G. Blackmond
J. Am. Chem. Soc.,
2017, 139, (27), 9238-9245; DOI:10.1021/jacs.7b03716
06/2017
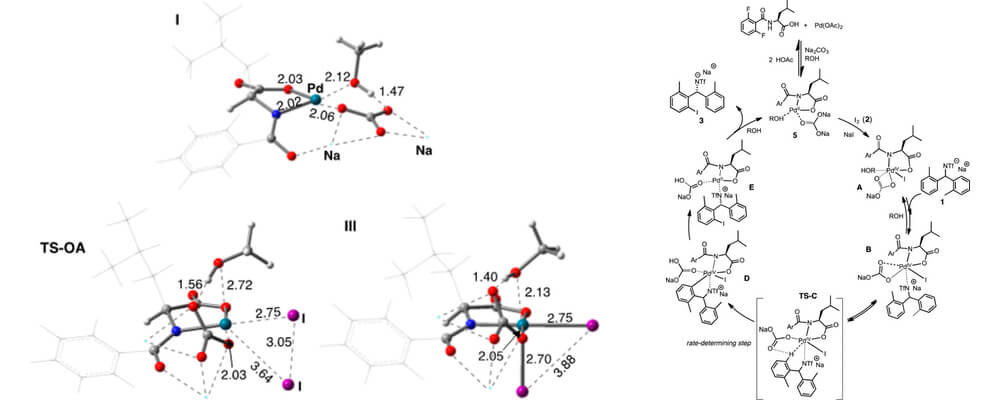
This collaborative report from the Blackmond, Yu, Sigman and Musaev groups builds upon work within the Center that focuses on elucidating the role of monoprotected amino acid ligands in the palladium-catalyzed asymmetric C–H activation chemistry.
Kinetic and mechanistic studies of the desymmetrization of benzhydrylamine using Pd/monoprotected amino acid ligands (Pd/MPAA) via C–H functionalization with molecular iodine provide mechanistic insight into the rate-determining step and the oxidation state of Pd in the C–H functionalization step.
Enantiomeric excess is strikingly insensitive to temperature from ambient temperature up to over 70 °C, and reaction rate is insensitive to the electronic characteristics of the ligand’s benzoyl protecting group. The reaction is highly robust with no evidence of catalyst deactivation. Intriguingly, C–H bond breaking does not occur prior to the addition of I2 to the reaction mixture. Electrochemical experiments demonstrate the viability of oxidative addition of I2 to Pd(II). Together with 19F NMR studies, these observations suggest that iodine oxidizes Pd prior to addition of the amine substrate.
This work may lead to a better general understanding of the subtle variations in the reaction mechanisms for C–H functionalization reactions that may be extant for this ligand class depending on substrate, amino acid ligand and protecting group, and reaction conditions.