A General Method for Artificial Metalloenzyme Formation through Strain-Promoted Azide–Alkyne Cycloaddition
Hao Yang, Poonam Srivastava, Chen Zhang, and Jared C. Lewis
Chem. Bio. Chem.,
2014, 15, 223-227; 10.1002/cbic.201300661
12/2013
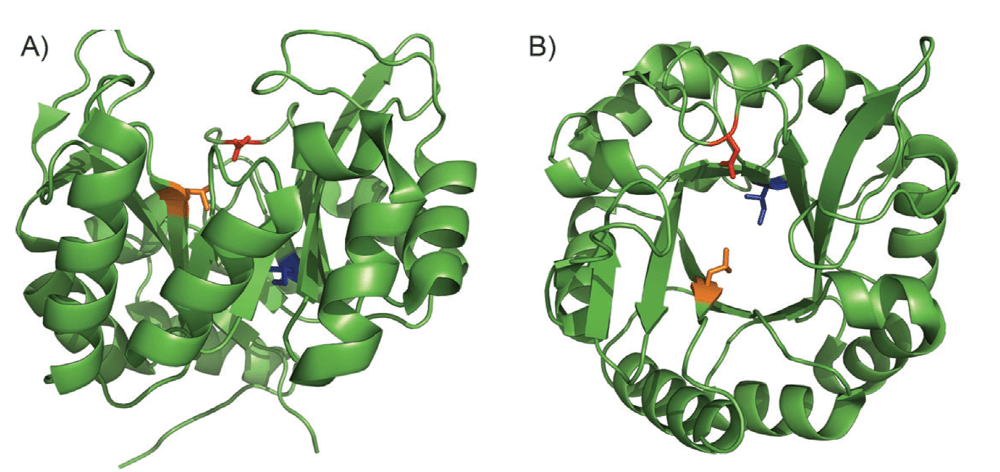
The Lewis group have an ongoing program exploring the preparation and application of Artificial Metalloenzymes (ArM’s) in novel transformations, such as C–H Functionalization.
In this report they describe a rapid, general and efficient synthesis of ArM’s using the strain-promoted azide-alkyne cycloaddition (SPAAC) reaction, generating these catalysts from scaffold proteins containing a p-azido-L-phenylalanine residue. The efficiency of the reaction is such that the desired catalysts are produced even in the presence of reactive residues or motifs, such as cysteine or reactive components of cellular lysate.
The general scope of this method with respect to both the protein scaffold and cofactor means that any desired metal complex can be used in combination with unique protein scaffolds, demonstrating broad utility.
The power of these new systems was demonstrated with a dirhodium complex embedded in the protein showing catalytic decomposition of diazo compounds and application in Si–H functionalization and olefin insertion.