Effect of Molecular Structure Perturbations on the Performance of the D−A−π−A Dye Sensitized Solar Cells
X. Kang, J. Zhang, D. O’Neil, A. J. Rojas, W. Chen, P. Szymanski, S. R. Marder, and M. A. El-Sayed
Chem. Mater.,
2014, 26, 4486-4493
07/2014
A collaborative project between the Marder and El-Sayed groups explores the effect of molecular variation in the performance of a series of D-A-Pi-A dyes, prepared through C–H functionalization, on their solar to electric power conversion efficiencies.
Organic solar dyes typically contain electron-rich (donor: D) and electron-poor (acceptor: A) regions connected by a conjugated bridge, with a recent advance being the inclusion of an auxiliary electron-withdrawing unit into the bridge to facilitate electron transfer.
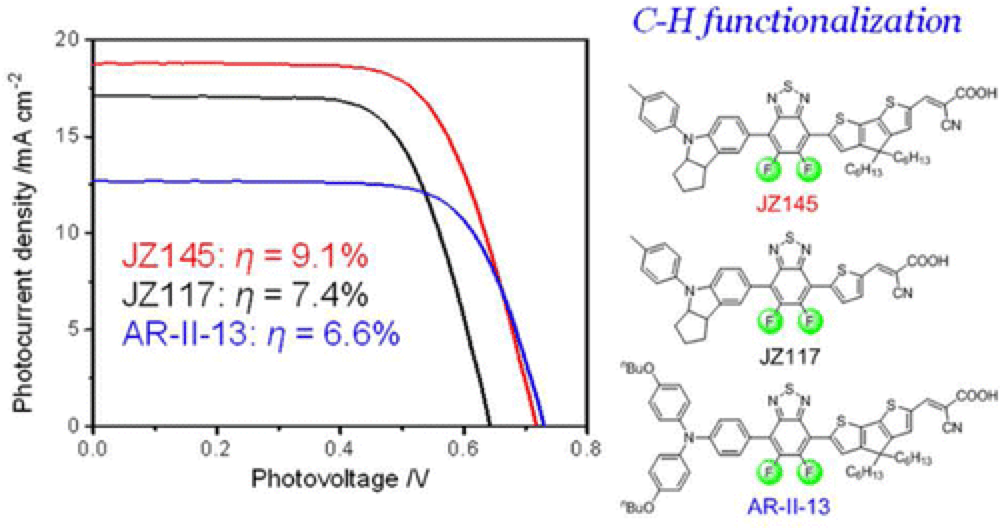
This work builds upon the discovery that C–H Functionalization provides a direct and efficient method for the elaboration of the electron-withdrawing unit, the 5,6-difluoro-2,1,3-benzothiadiazole (DFBTD), leading to the construction of novel D-A-Pi-A sensitizers, incorporating a new bridging system based on the cyclopentadithiophene (CPDT) unit, the side-chains of which were designed to reduce dyeaggregation.
This work describes the synthesis and evaluation of four novel dyes, providing a clear picture of how the different molecule variations impact the efficiencies of these systems. Incorporation of the electron-withdrawing DFBTD unit and bridging CPDT led to a dye with a solar to electric power conversion efficiency of 9.1%.
Investigations are ongoing to explore the new electron deficient units that can be functionalized using C–H activation, typically a difficult reaction using more conventional cross-coupling technologies.