Application of a Palladium-Catalyzed C−H Functionalization/Indolization Method to Syntheses of cis-Trikentrin A and Herbindole B
Raul A. Leal, Caroline Bischof, Youjin V. Lee, Shota Sawano, Christopher C. McAtee, Luke N. Latimer, Zachary N. Russ, John E. Dueber, Jin-Quan Yu, Richmond Sarpong
Angew. Chem. Int. Ed.,
2016, 128, 39, pp 12003-12007; DOI: 10.1002/ange.201605475
08/2016
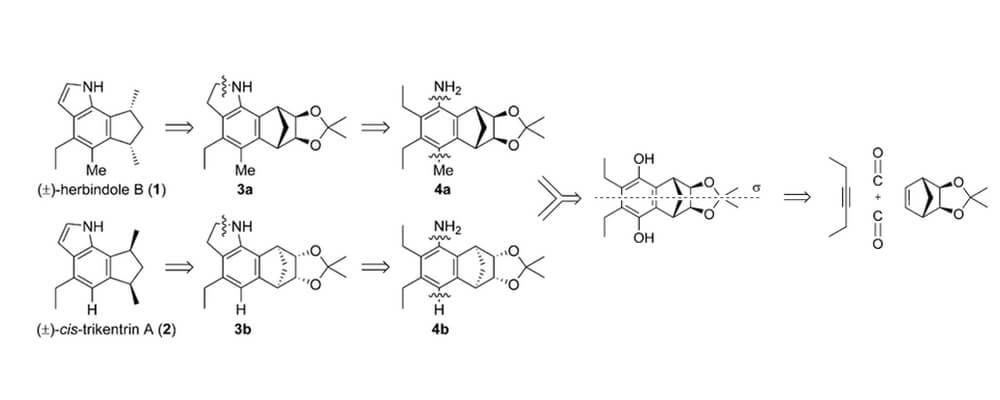
The Sarpong and Yu groups describe their collaborative strategy for the formal synthesis of the cis-trikentrin A and herbindole B natural products.
The Sarpong group identified a ruthenium catalyzed [2+2+1+1] cycloaddition, previously unused in natural product synthesis, that rapidly constructed the core of these natural product families. A Buchwald-Hartwig amination was used to introduce the aniline functionality at the sterically congested site.
Working with the Yu group a novel indole formation directly at an sp3 C–H bond was developed, furnishing the product in good yield. This procedure proved to be general for other substrates to furnish both the trikentrin and herbindole natural products.
The development of this new indole forming technology is already being incorporated into future strategies toward complex molecule synthesis thanks to the ubiquity of this structural motif in natural products.