An indacenodithiophene-based semiconducting polymer with high ductility for stretchable organic electronics
Yilin Li, Wesley K. Tatum, Jonathan W. Onorato, Sierra D. Barajas, Yun Young Yang and Christine K. Luscombe
Polym. Chem.,
2017, Advance Article; DOI:10.1039/C7PY00435D
05/2017
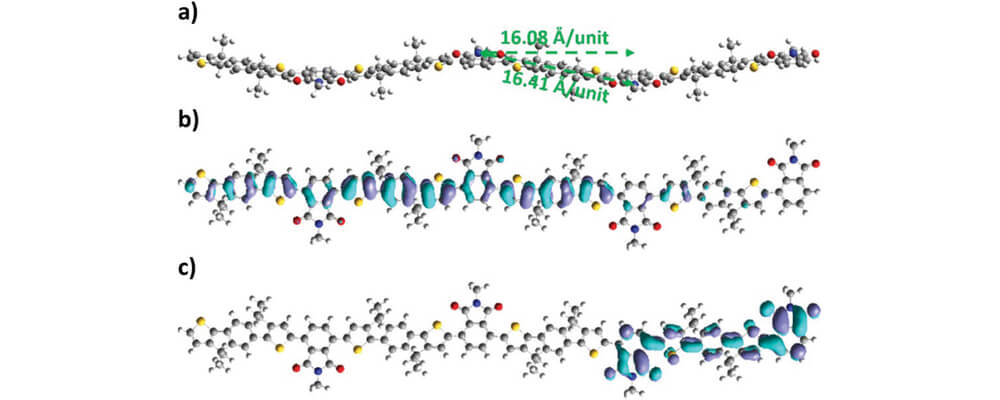
This work from the Luscombe Group describes tha application of C–H Functionalization technology to the synthesis of an alkyl-substituted indacenodithiophene-based donor–acceptor π-conjugated polymer (PIDTBPD), with low stiffness and high ductility.
The polymer was synthesized after DFT calculations predicted that it would have a kinked backbone conformation while showing strong intramolecular charge transfer (ICT), suggestive of the fact that it would be beneficial to the polymer's elasticity and charge mobility.
C–H Functionalization, or direct arylation polymerization (DArP), was used as a streamlined entry to this polymer.
Mechanical studies indicate that PIDTBPD has relatively rapid stress-relaxation properties, which lead to a low elastic modulus of 200 MPa and high crack-onset strain of ca. 40% (lower limit). A moderate charge carrier mobility of 2 × 10−3 cm2 V−1 s−1 with a current on/off ratio of 2.5 × 106 was obtained from the fabricated OFETs. Further experiments were performed to elucidate the structural aspects of this polymer.