Enantioselective C(sp3)‒H bond activation by chiral transition metal catalysts
Tyler G. Saint-Denis, Ru-Yi Zhu, Gang Chen, Qing-Feng Wu, Jin-Quan Yu
Science,
2018, 359, 6377; DOI:10.1126/science.aao4798
02/2018
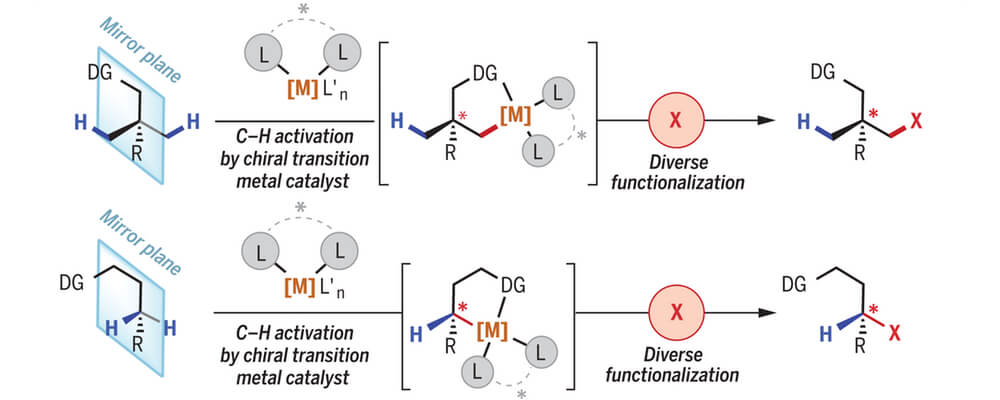
One sub-category of C–H functionalization is C–H activation, which involves the generation of a discreet organometallic intermediate (i.e. a species containing a transition metal–carbon bond) through the reactivity, (either stoichiometric or catalytic) between an unreactive C–H bond and a reactive transition metal catalyst. Though significant strides in regioselective C–H activation have been achieved over the past half century, considerable less attention has been focused on the development of robust enantioselective C(sp3)–H activation processes.
This review covers the history of ligand design and the corresponding stereomodels invoked in transition metal-catalyzed C(sp3)–H activation. Enantioselective C(sp3)–H activation, in its present state, may be divided into three broad categories: (A) point desymmetrization, which encompasses the generation of a stereocenter adjacent or distal to the C–H bond undergoing reactivity; (B) methylene desymmetrization, which includes the generation of a stereocenter on the same carbon undergoing C–H bond reaction; and (C) kinetic resolution, which entails the differentiation of enantiomers of a racemic mixture by a chiral catalyst, such that only one enantiomer reacts with the chiral catalyst. A variation of this C–H kinetic resolution is parallel kinetic resolution, in which each enantiomer is converted to different products by the same chiral catalyst.
The examples presented in the review should attest to the promise enantioselective C(sp3)–H activation has to enrich synthetic disconnections and expedite chemists’ synthetic endeavors.
Author: Tyler St Denis