Role of N-Acyl Amino Acid Ligands in Pd(II)-Catalyzed Remote C–H Activation of Tethered Arenes
Gui-Juan Cheng, Yun-Fang Yang, Peng Liu, Ping Chen, Tian-Yu Sun, Gang Li, Xinhao Zhang, K. N. Houk, Jin-Quan Yu, and Yun-Dong Wu
J. Am. Chem. Soc.,
2014, 136, 894-897; 10.1021/ja411683n
12/2013
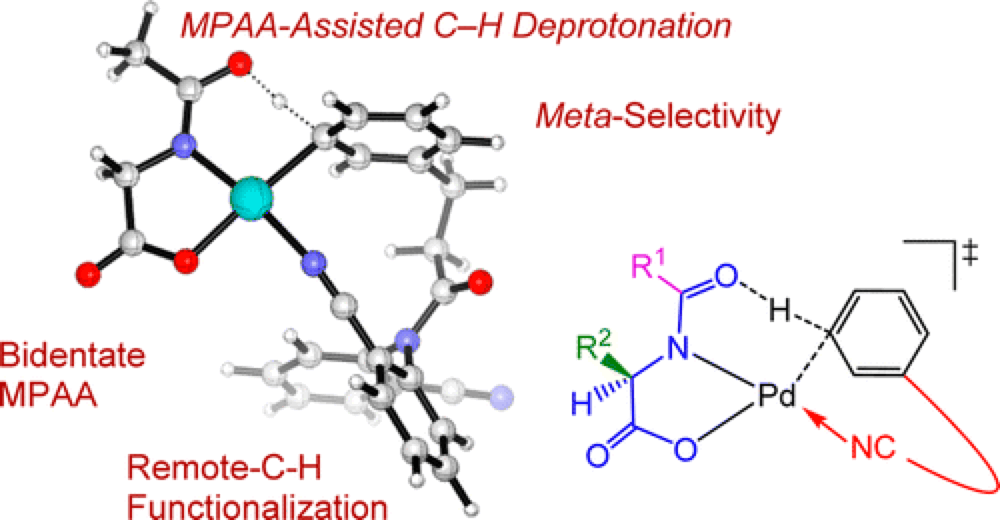
Control over the selective functionalization of the positions around an aromatic ring is one of the leading goals of the Center, with a myriad of applications in both the pharmaceutical and materials sciences. The approach adopted by the Yu group is to use a template with a weakly binding directing group to dictate the site of Pd-catalyzed C–H functionalization.
A key recent advance is the development of a template that favors functionalization at the meta-position. This report, a collaboration between the Yu group at Scripps, the Houk group at UCLA and the Wu group at Peking, delves deeper into this mechanism to learn more about the details that control this selectivity and use this to inform future designs of the templates.
This study sheds light on some of the features central to the N-acyl amino acid/Pd(II)-catalyzed C–H Functionalization developed by the Yu group and details some of the key considerations that need to be accounted for when designing templates to direct C–H Functionalization.