Directed evolution of RebH for catalyst-controlled halogenation of indole C–H bonds
Mary C. Andorfer, Hyun June Park, Jaylie Vergara-Colla and Jared C. Lewis
Chemical Science,
2016, 7, 3720-3729; 10.1039/C5SC04680G
02/2016
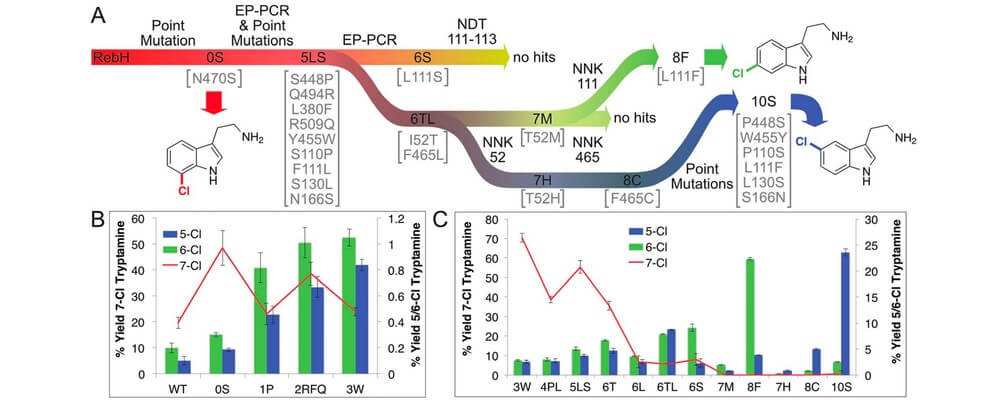
Arguably the key challenge in C–H functionalization is selecting between the multitude of C–H bonds present in a typical organic molecule. The Center is exploring a number of strategies to assert control in these reactions.
This report from the Lewis group describes the directed evolution of rebeccamycin halogenase (RebH), that results in three engineered halogenases that can reliably select for the ortho-, meta- and para-positions of indole substrates for C–h halogenation.
This transformation is possible employing molecular oxygen as the oxidant, halide salts as the halogen source and under physiological conditions.
The generality of the evolution strategy used in this study and the selectivity assay developed suggest that the selectivity of other enzymes could be evolved in a similar fashion that would enable a whole suite of non-directed C–H functionalization reactions.
