Synthesis of 8-Aminoquinolines by Using Carbamate Reagents
Donghyeon Gwon, Heejun Hwang, Hye Kyung Kim, Seth R. Marder, Sukbok Chang
Chem. Eur. J.,
2015, 21, 48, 17200-17204; 10.1002/chem.201503511
10/2015
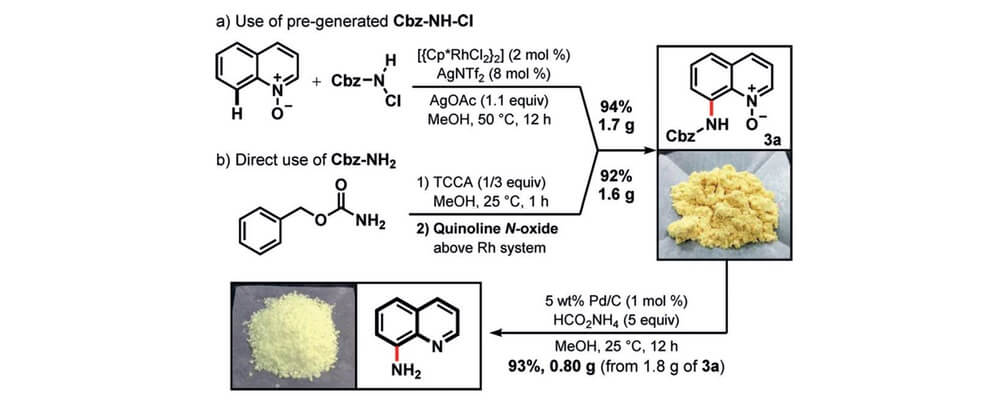
This collaborative project was driven by our international SAVI program, the VIC–HF. This report from the Marder (GA Tech, USA) and Chang groups (KAIST, South Korea), describes a rapid and convenient entry into 8-aminoquinolines, useful structural motifs in a variety of applications, from LCD displays to catalyst ligands and natural products.
Using a Rh(III)-catalyzed approach this scalable synthesis was performed on the multigram scale. Using a carbamate C–H amination followed by facile deprotection directly furnished the free amine. The reaction was found to be general for variously substituted substrates and could be performed with as low as 2 mol% of the Rh(III)-catalyst and 1 mol% of the Pd/C for the deproctection step.
This procedure is both straightforward and capable of providing valuable structural skeletons.