Ligand-accelerated enantioselective methylene C(sp3)–H bond activation
Gang Chen, Wei Gong, Zhe Zhuang, Michal S. Andrä, Yan-Qiao Chen, Xin Hong, Yun-Fang Yang, Tao Liu, K. N. Houk, Jin-Quan Yu
Science,
2016, 353, 6303, 1023-1027; DOI:10.1126/science.aaf4434
09/2016
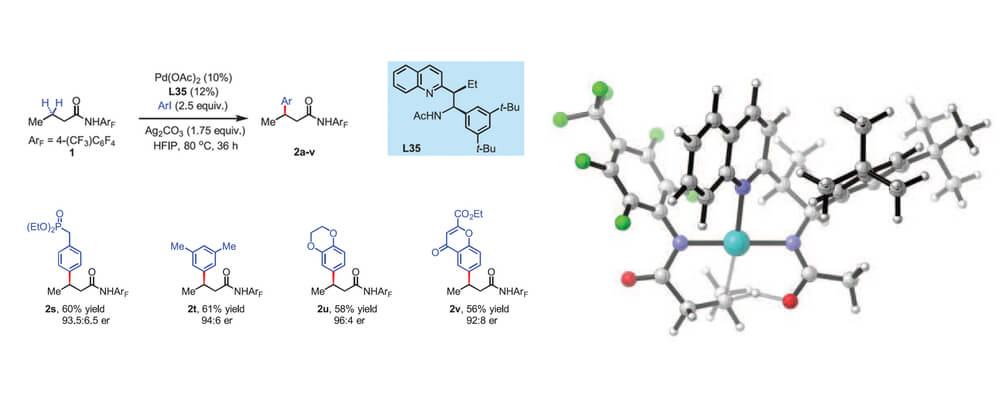
This collaborative report from the Yu and Houk groups is the culmination of many years work in the Yu group and opens news vistas for C–H activation chemistry.
The Yu group has led the field of using weakly coordinating directing groups to functionalization C(sp2)–H bonds. Teams within the Center have helped provide a better understanding of this reaction, which has led to the development of systems based on copper-catalysis and the development of ligand-accelerated reactions that overcome the inherent ‘background’ reaction and provide exquisite levels of control.
This ligand acceleration is key to this development, being able to selectively functionalize the C–H bond at a beta-position to an amide or carboxylate in a enantioselective fashion. Behind this project there is a vast amount of ligand development and understanding that has enabled this transformation.
This represents the first place-marker for a whole strategy for controlling selectivity that will form a primary focus for the Center moving forward.