Bimodal Evans–Polanyi Relationships in Dioxirane Oxidations of sp3 C–H: Non-perfect Synchronization in Generation of Delocalized Radical Intermediates
Fengjiao Liu, Zhongyue Yang, Yanmin Yu, Ye Mei, and K. N. Houk
J. Am. Chem. Soc.,
2017, 139 (46), pp 16650–16656; DOI:10.1021/jacs.7b07988
10/2017
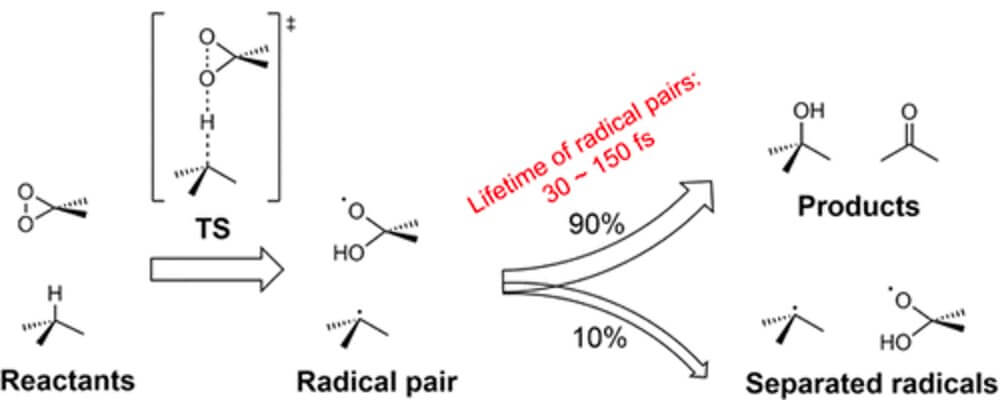
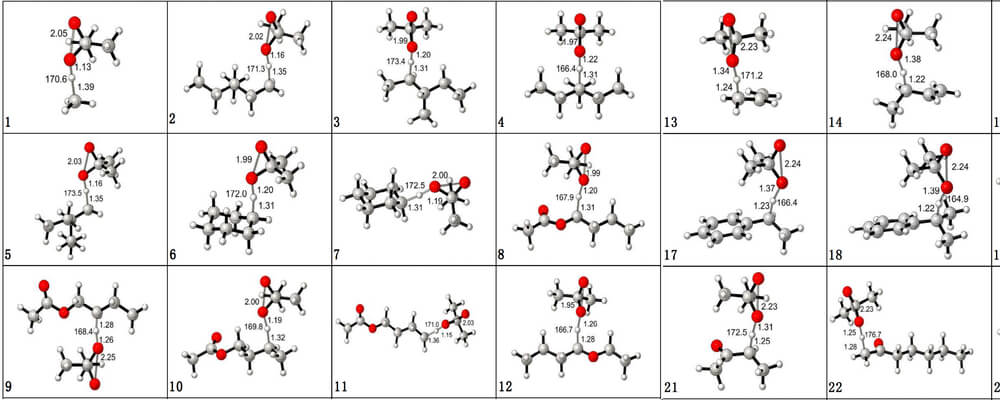
In this collaborative report from the Houk group, at UCLA, and the Mei group, at East China Normal University, Shanghai, the authors describe their investigations into fundamental substrate factors that influence reactivity and site-selectivity in sp3 C–H Oxidations. Their findings are predicted to be applicable across all reactions involving the breaking of C–H bonds.
Using Dimethyldioxirane (DMDO) and methyl(trifluoromethyl)-dioxirane (TFDO) C–H oxidation as a model reaction for this study, since these are outstanding reagents for selective oxidation of a wide variety of alkanes under mild conditions in the absence of metal catalysts, a “baseline” of the factors that impact the breaking of a C–H bond was composed.
By investigating the origins of reactivity and selectivity for this class of reaction, the authors are able to identify trends that will inform other C–H functionalization technologies.