Organocatalytic C–H hydroxylation with Oxone enabled by an aqueous fluoroalcohol solvent system
Ashley M. Adams and J. Du Bois
Chemical Science,
2014, 5, 656-659; 10.1039/C3SC52649F
12/2013
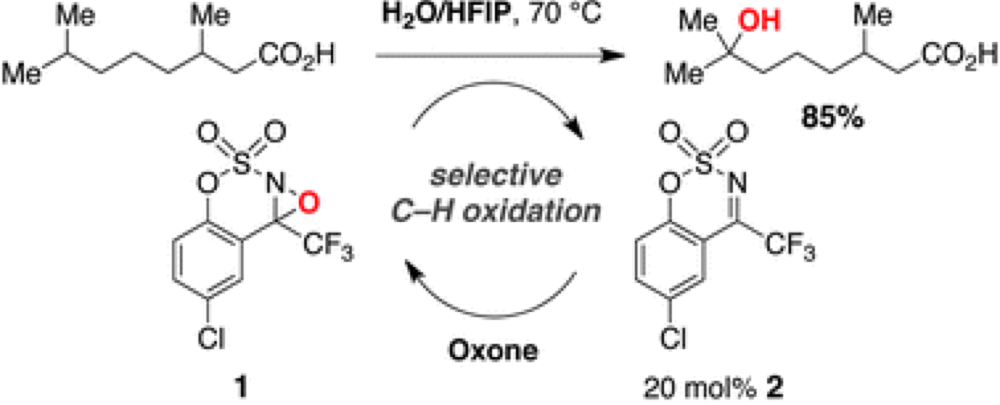
The development of methods for the selective hydroxylation of C–H bonds has revolved around transition-metal catalysis. Some recent findings from the Du Bois group have opened the door to using organocatalysis to selectively oxidize tertiary and benzylic C–H bonds in a catalytic fashion.
Initial studies in this area found that the reaction suffered from poor conversion, though with significant levels of selectivity. Through some detailed control experiments it was established that the presence of water and a build-up of the alcoholic product promote decomposition of the oxaziridine organocatalyst. However, after a logic guided solvent screen, a biphasic system was found to be optimal, using water with a fluoroalcohol solvent. It is proposed that the fluorous phase forms micro-droplets, in which the effective concentration of the organic reagents is increased, greatly amplifying the rate of reaction and maintaining the integrity of the catalyst and allowing the use of Oxone as a terminal oxidant.
These findings not only increase the scope and efficacy of this reaction but also lay the foundations for numerous other catalytic C–H functionalizations based around these reaction conditions, making reaction in aqueous media a tantalizing possibility.