Rapid Construction of a Benzo-Fused Indoxamycin Core Enabled by Site-Selective
C−H Functionalizations
T. Aaron Bedell, Graham A. B. Hone, Damien Valette, Jin-Quan Yu, Huw M. L. Davies, Erik J. Sorensen
Angew. Chem. Int. Ed.,
2016, 55, 29, 8270–8274; 10.1002/anie.201602024
05/2016
One of the core drives within the Center is the development of novel transformations and their application in the synthesis of complex molecules. While a novel transformation may look straightforward and efficient on a specific set of selected substrate it is only when they are applied in the synthesis of complex systems that many of the challenges become apparent and the reliability of the transformation is truly tested.
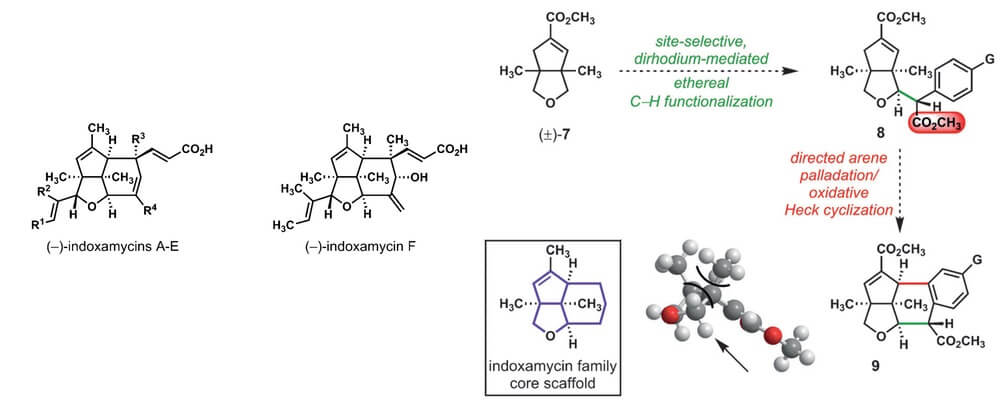
This report describes a project designed and driven by the Sorensen group that challenges transformations developed in the CCHF in the development of a strategy that significantly streamlines the synthesis of an important class of natural products, the indoxamycins.
The synthetic strategy devised employs three distinct C–H Functionalization events to stitch together the carbon framework, a central donor/acceptor carbene C–H insertion that sets the stereochemistry of the molecule, an ester directed oxidative Heck cyclization and an opportunity for a late-stage vinylation.
Find out more about how the Sorensen, Davies and Yu groups worked together to streamline the synthesis of the core of this natural product family.