Complex Hexacyclic Compounds via a Tandem Rh(II)-Catalyzed Double-Cyclopropanation/Cope Rearrangement/Diels–Alder Reaction
Jillian E. Spangler, Yajing Lian, Sandeep N. Raikar and Huw M. L. Davies
Organic Letters,
2014, 16, 18, 4794-4797; 10.1021/ol502257d
09/2014
The Davies Group reports an unusual cascade sequence that demonstrates the synthetic potential that can be unlocked when you understand the factors that control the reactivity of dirhodium-carbenes.
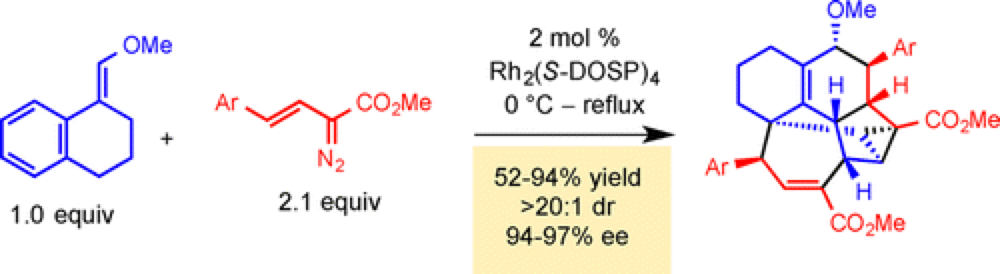
During theory-led investigations into the scope and limitations of the combined C–H Functionalization / Cope-rearrangement, a reaction previously established by the Davies group, a new reaction pathway was discovered that provides rapid and stereo-controlled access to a complex hexacyclic framework containing ten stereocenters in upto 97% ee.
By designing the steric environment of the substrate such that the initial C–H Functionalization is unfavored, this new cascade pathway takes over. With appropriately configured substrates this is a general reaction, providing a facile and rapid entry into very complex scaffolds potential advanced intermediates toward natural products and API's.
This is a further demonstration of how a detailed understanding of these reactions can provide new transformations and modes of reactivity.