Silylation of C–H bonds in aromatic heterocycles by an Earth-abundant metal catalyst
Anton A. Toutov, Wen-Bo Liu, Kerry N. Betz, Alexey Fedorov, Brian M. Stoltz and Robert H. Grubbs
Nature,
2015, 518, 80-84; 10.1038/nature14126
01/2015
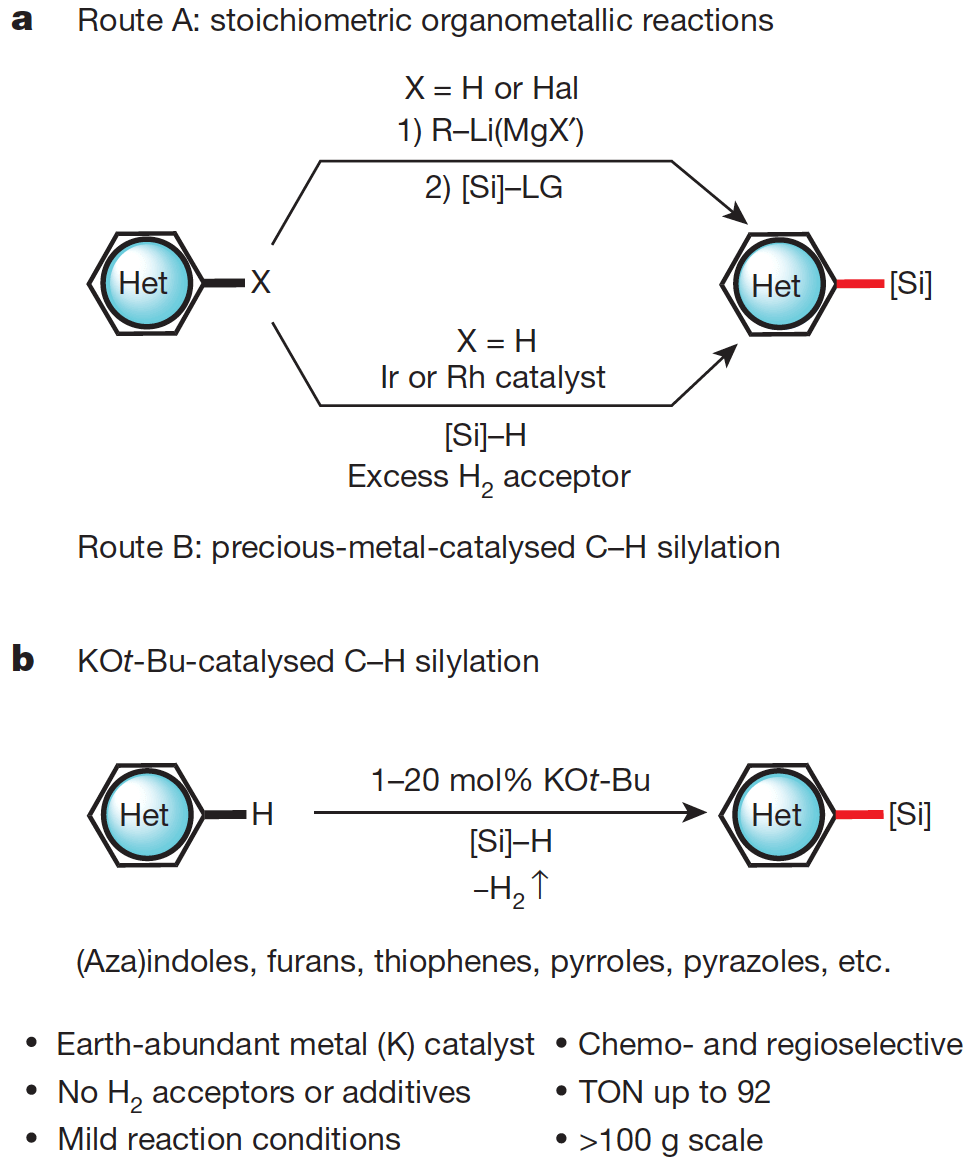
This report describes the results of a collaborative project between the Stoltz and Grubbs groups at Caltech. Together they have developed an efficient, general and robust method for the C–H silylation of heterocyclic compounds, key molecular motifs in pharmaceutical agents and materials science.
This transformation generally relies upon precious metal catalysts, based on palladium, platinum or iridium, however this new system employs potassium tert-butoxide as a catalyst, an in-expensive and earth abundant commodity.
Not only is the reaction general in its scope, it is also readily scalable, with no demonstrated sensitivity to moisture or atmospheric oxygen. This was demonstrated with the reaction performed on 150g scale.
The collaboration was highlighted on the homepage of Caltech this month.
Carbon–Silicon bonds can be readily transformed to a broad range of functional group, thus this transformation represents a rapid and sustainably entry into the facile functionalization of heterocyclic compounds.
Studies are currently underway to understand better this new catalytic manifold and learn lessons from this new system to help design new C–H Functionalization reactions in the future.
Related Content
-

08/2015
2015 GSIA
OUTREACH
-

04/2015
Elementary 19 Documentary
OUTREACH